Cathode materials with octahedral coordination are currently the most widely used type of cathodes. Among them, materials that feature anion (oxygen) redox, such as lithium-rich manganese-based cathodes and lithium-rich rock-salt phase materials, exhibit specific energies above 250 mAh/g, making them strong candidates for next-generation cathodes. However, these materials often face challenges such as oxygen loss and structural collapse at high voltages. Even with modifications such as doping, these problems cannot be fundamentally resolved. In recent years, many cutting-edge studies have focused on controlling the cathode’s microscopic structure to significantly enhance electrochemical performance. Research indicates that altering the coordination environment and atomic arrangement can fundamentally impact electrochemical behavior.
Amorphous materials—a broad class of materials without periodic crystal structures—can also serve as electrode materials. Their disordered atomic arrangements often endow them with unique properties. Traditionally, research has focused on crystalline cathodes, analyzing performance, mechanisms, and modifications, and it has been assumed that anion redox occurs only in octahedrally coordinated crystalline cathodes. Studies on amorphous cathodes, however, remain limited. Recent research shows that amorphous structures can change lithium extraction mechanisms, offering ultra-high specific energies and excellent electrochemical stability.
To this end, Professor Dingguo Xia’s team at Peking University reported a tetrahedrally coordinated amorphous Li-V-O-F cathode. Through in-depth investigation of its electrochemical behavior and charge-compensation mechanisms, the team discovered that it can activate an “O–O dimer” type anion redox reaction at relatively low voltages. Remarkably, this reaction occurs without oxygen loss, voltage decay, or structural instability. This finding revises previous understanding and highlights the significance of amorphous materials as promising candidates for next-generation electrodes. The work, titled “An amorphous Li-V-O-F cathode with tetrahedral coordination and O–O formal redox at low voltage”, was published in Nature Materials (https://rdcu.be/exjDA).
The study found that after amorphization, the Li-V-O-F cathode exhibits a small oxidation plateau at around 4.1 V (Figures 1a, b). Compared with the non-amorphized (crystalline) Li-V-O-F cathode, the amorphous structure forms new coordination pairs at 1.3–1.5 Å after charging, corresponding to O–O dimers (Figure 1c). Coordination analysis further shows that the tetrahedral coordination environment is preserved in the amorphous structure even after charging (Figure 1d).

Figure 1. Evolution of the macroscopic structure and coordination environment of the Li-V-O-F cathode.

Figure 2. Redox mechanisms of the Li-V-O-F cathode during charge and discharge.
X-ray absorption spectroscopy was used to probe the electronic states of V and O ions during charge and discharge. It was found that upon charging to high voltages, the V–O hybridized hole states increase, indicating oxidation of the oxygen species (Figures 2b, c). In contrast, the V ions show a reduction in valence (Figure 2d), demonstrating that the high-voltage oxidation is compensated by oxygen, with the V ions exhibiting the characteristic coupling effect of anion redox.

Figure 3. mRIXS results of oxygen at different electrochemical stages.

Figure 4. Molecular dynamics simulations of the crystalline and amorphous structures.
Resonant inelastic X-ray scattering (RIXS) was employed to investigate the redox behavior of oxygen in depth. In the amorphous structure during charging, new fingerprint signals appear in the oxygen mRIXS spectra (Figures 3b, d), corresponding to the formation of peroxide-like O–O bonds. These signals disappear reversibly upon discharge (Figure 3c), demonstrating the reversibility of anion redox in the amorphous Li-V-O-F structure.
Ab initio molecular dynamics simulations were performed to study the structural evolution of Li-V-O-F cathodes after lithium extraction in both crystalline and amorphous structures. In the crystalline structure, oxygen atoms remain fixed within the lattice framework, and even when subjected to high temperatures of 1000 K to promote reactions, the structure shows no significant change (Figures 4a, d, g). In contrast, under the same conditions, the amorphous structure undergoes diverse structural changes, with ions rearranging in a disordered manner, enabling active oxygen to more easily overcome thermodynamic barriers and form stable O–O dimers (Figures 4b, c, e, f, h). These computational results are in excellent agreement with the experiments, highlighting the intrinsic structural features and thermodynamic favorability of the amorphous structure in activating O–O dimer redox reactions.
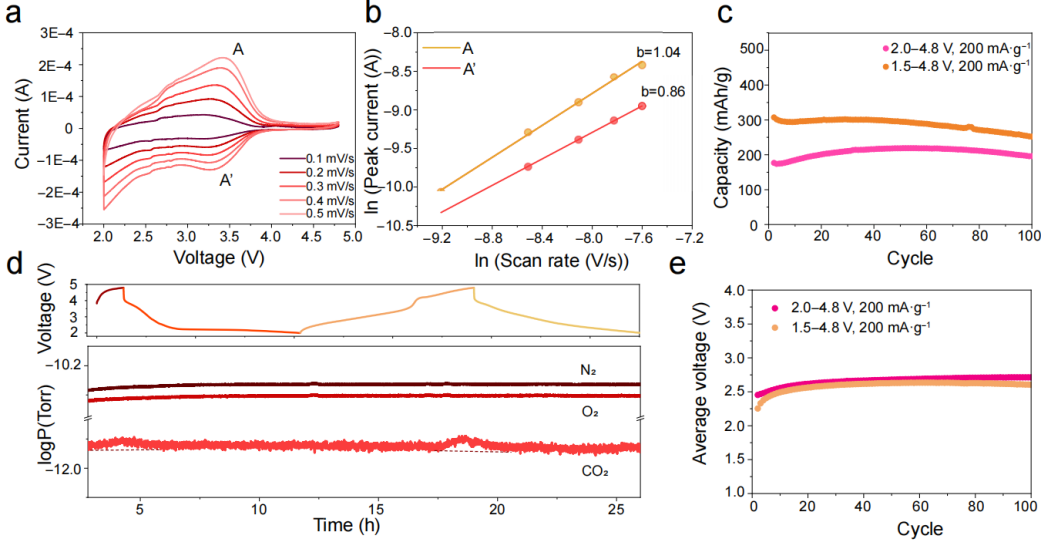
Figure 5. Electrochemical characteristics of the amorphous Li-V-O-F cathode.
Cyclic voltammetry demonstrated that the electrochemical reactions in the amorphous Li-V-O-F cathode are primarily pseudocapacitive (Figures 5a, b). This behavior arises from the amorphous structure, which creates numerous nanoscale channels that facilitate rapid lithium-ion transport. The amorphous Li-V-O-F cathode delivers a high specific capacity of over 300 mAh/g in the voltage range of 1.5–4.8 V (Figure 5c), and importantly, shows no oxygen evolution at high-voltage charging (Figure 5d) or voltage decay (Figure 5e). These electrochemical features demonstrate the excellent stability and structural reversibility of the anion redox reactions.
This work, combining advanced characterization techniques with theoretical calculations, reveals a novel anion redox mechanism in tetrahedrally coordinated amorphous cathodes, opening new directions for the development of high-energy-density cathode materials.
Ph.D. student Kun Zhang from the School of Materials Science and Engineering (PKU) is the first author, and Professor Dingguo Xia is the sole corresponding author. This work was supported by the National Key R&D Program of China and the National Natural Science Foundation of China.
DOI: 10.1038/s41563-025-02293-9